FDA Approves BAVENCIO® (avelumab) Plus INLYTA® (axitinib) Combination for Patients with...
ROCKLAND, Mass. and NEW YORK, May 14, 2019 /PRNewswire/ -- BAVENCIO is the first anti-PD-L1 in combination with INLYTA approved by FDA for first-line treatmen
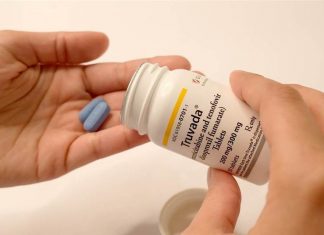
Gilead to donate HIV prevention drug Truvada to 200K uninsured patients
Gilead Sciences announced that it will provide free Truvada as PrEP for 200,000 people through 2030. Activists say Gilead is moving to protect Truvada revenue.
Takeda Simplifies Portfolio and Accelerates Deleveraging Through Two Divestitures | Business...
Takeda Pharmaceutical Company Limited (TSE:4502/NYSE:TAK) (“Takeda”) today announced that it has entered into agreements to divest its Xiidra® (lifite
U.S. FDA Approves VYNDAQEL® and VYNDAMAX™ for Use in Patients with...
Pfizer Inc. (NYSE:PFE) announced today that the U.S. Food and Drug Administration (FDA) has approved both VYNDAQEL® (tafamidis meglumine) and VYNDAMAX
AveXis Presents Data Showing Efficacy Of Zolgensma In Broad Spectrum Of...
AveXis, a Novartis company, announced interim data from ongoing trials of the investigational product Zolgensma or onasemnogene abeparvovec-xioi; AVXS-101 that showed positive results across a broad spectrum of patients with spinal muscular atrophy or SMA.
ViiV Healthcare Submits New Drug Application to US FDA for the...
ViiV HC submits New Drug Application to US FDA for first monthly, injectable, two-drug regimen of cabotegravir and rilpivirine for treatment of HIV
Plaque Psoriasis Armamentarium Gets New Addition | Markets Insider
(RTTNews) - The therapeutic armamentarium for plaque psoriasis patients is fairly well-stocked, and now there is one more addition to it.The FDA ha
Boston Scientific Receives FDA Approval for LOTUS Edge™ Aortic Valve System
MARLBOROUGH, Mass., April 23, 2019 /PRNewswire/ -- Boston Scientific Corporation (NYSE: BSX) today announced it has received U.S. Food and Dru
TB Alliance and Mylan Announce Global Collaboration to Commercialize Investigational Drug...
NEW YORK and HERTFORDSHIRE, England and PITTSBURGH, April 18, 2019 /PRNewswire/ -- Non-profit drug developer, TB Alliance, and pharmaceutical company
Gilead and insitro Announce Strategic Collaboration to Discover and Develop Novel...
Gilead Sciences Inc. (Nasdaq: GILD) and insitro announced today that the companies have entered into a strategic collaboration to discover and develop